Background: Fms-like tyrosine kinase 3 internal tandem duplication (FLT3-ITD) is a well-established prognostic marker in acute myeloid leukemia (AML), whereas the impact of mutations in the Tirosine-Kinase Domain (TKD) is controversial. The association of TK inhibitors (TKIs) with intensive chemotherapy improved the overall survival (OS) of FLT3-mutated AML patients in randomized clinical trials and is recommended by the European LeukemiaNet panel of experts. Nevertheless, TKIs are not available for most patients treated in Low- and Middle-Income Countries (LMIC) and there is a scarcity of data concerning the outcome of treatment in these resource-constrained settings nowadays.
Methods: This is a real-world study encompassing newly diagnosed adults with FLT3-mutated AML intensively treated at public hospitals in Brazil between January 2015 and December 2020, who were registered in the International Consortium on Acute Leukemias (ICAL) ICAML2015 Study. No patient received TKIs as part of the treatment. FLT3 mutation was tested in diagnostic samples by fragment analysis method. We used the Adapted Genetic Risk (AGR) score reported by Silveira et al. (2020), which is an adaptation of the European Leukemia Net (ELN) 2017 classification. The primary endpoints were event-free survival (EFS) and OS, calculated by Kaplan-Meier method. Relapse rate was calculated through competing risk analysis.
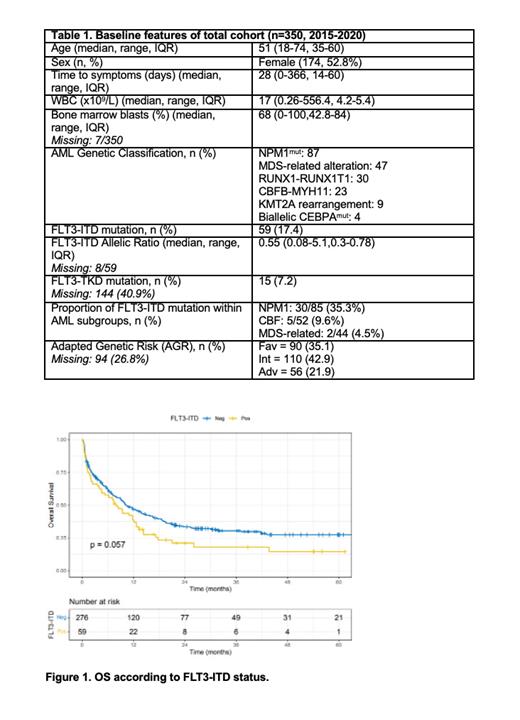
Results: Overall, 350 pts (median age: 51y; 52.8% females) were included. FLT3-ITD and -TKD mutations were detected in 17.5% and 7.2% of the patients, respectively. Ninety patients (35.1%) were classified in the favorable risk group, 110 (42.9%) in the intermediate, and 56 (21.9%) in the adverse risk group, according to the AGR. The most frequent co-mutation was in the NPM1 gene. The remaining features are summarized in Table 1. Seventy-eight patients underwent allogeneic stem-cell transplantation (HSCT) in first complete remission (22.2%), of whom 27 had FLT3-ITD (i.e. 43.9% of patients harboring FLT3-ITD underwent HSCT). With a median follow-up period of 3.5 years, the median 4-year OS rate was 24.2% (95% CI 19.8-30.1), and the 4-y EFS 22.9% (95% CI 18.6-28.2). When only patients with FLT3-ITD mutations were analyzed, the 4-y OS rate was 14.4% which was inferior to that of patients with wild-type FLT3 (27.5%; p=0.057) (Figure 1). The cumulative incidence of relapse at four years was 39.4% for patients with FLT3-ITD and 43.9% for those with wildtype FLT3 (p=0.5), while the non-relapse mortality (NRM) at four years was 28.1% and 32.4%, respectively. The four-year OS rate for patients with FLT3-ITD mutations who underwent HSCT was 49.1% (95% CI 38.9-62.1). Patients with FLT3-ITD were further subdivided according to the allelic ratio (AR) in those with low (<0.5) and high (³ 0.5), and no significant difference in OS was detected between the two groups. Of note that patients harboring FLT3-TKD mutations showed a better overall survival rate compared to those with wild-type FLT3 (HR 0.49, 95% CI 0.26-0.93, p=0.029), but this result needs to be interpreted cautiously due the small size of the cohort.
Conclusion: our results show that the frequency of FLT3-ITD mutations and their association with worse prognosis was similar to those described in high-income countries previously to the introduction of TKI as the standard of care. Therefore, allogeneic HSCT remains a valid consolidation strategy for FLT3-mutated patients. However, less than half of the patients for whom HSCT was indicated were actually transplanted. Additionally, the allelic ratio of FLT3-ITD should no longer be considered a prognostic marker for intensively treated patients not receiving FLT3 inhibitors.
Disclosures
Silva:Pfizer: Honoraria; Libbs: Research Funding; Servier: Honoraria. Traina:Novartis: Consultancy. Duarte:Astrazeneca: Speakers Bureau. Rego:Abbvie: Honoraria, Speakers Bureau; Astellas: Research Funding, Speakers Bureau; Pfizer: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal